|
1/12/2024 0 Comments Aluminum atomic structureThe answer lines in the behavior of the molecular orbitals and the resulting bonds they can produce. So why do some elements behave as metals and others do not? For example, graphite conducts electricity, but is not metallic in other ways - it is not malleable - and can’t be heated and molded into other shapes. The hardness of a (solid) metal depends on how well its atoms packed together and how many electrons are contributed to the valence band of orbitals. This is quite different from other substances (such as diamond or water). Metals can often be slowly deformed into different shapes without losing their structural integrity or electrical conductivity – they are malleable! They can be melted (increased atomic movement), become liquid, and then allowed to cool until they solidify throughout this process, they retain their integrity and their metallic properties - they continue to conduct electricity. When a piece of metal is put under physical stress – for example it is stretched or deformed, the atoms can move relative to one another, but the electrons remain spread throughout the structure. There are no discrete bonds in this type of structure. In this model, the atomic cores are packed together and are surrounded by a cloud of electrons that serve as the “glue” that binds them together. Since the numbers of electrons entering and leaving are the same,the piece of metal remains uncharged. Metalstypically have high conductivity due to the ease with which electrons can movefrom one MO to another, and the fact that each MO extends throughout the wholepiece of metal. Electrical conductivity isessentially a measure of how easily electrons can flow through a substance. When an electrical potential 80 is applied acrossthe metal, electrons from an external source can easily enter the valence bandand electrons can just as easily leave the metal. While nuclei and core electrons remain more or less locked in position,the valence electrons can “spread out” forming a kind of “electronsea” within the metal. 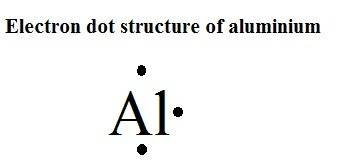
The physical consequences of this are that the valence electronscan move relatively easily from MO to MO, since their energies are very closetogether. Consider aluminum: it has threevalence electrons and in the ground (lowest energy) state has an electron configurationof 3s2 3p1, this implies that it has two unoccupied 3p orbitals (although whatthe meaning of an unoccupied orbital is not completely clear.) We can considerthe bonding molecular orbitals in aluminum to be formed from all the availableatomic orbitals - which means that there are many bonding MOs that are not occupiedby electrons. This is because in metals there are typicallyfewer electrons than there are atomic orbitals. Moreover,in metals the bonding molecular orbitals (known as the valence band) are ableto accommodate more electrons. The energy gap between the bonding and anti-bonding orbitalsis called the band-gap, and in a metal this band-gap is quite small (recall thatthe gap between the bonding and antibonding MOs in diamond is very large). As moreand more MOs are formed - the energies between them will get smaller and smallerand for a macroscopic piece of metal (that is one you can see) for all intentsand purposes the energy gap between the individual bonding MOs will be negligible.These orbitals produce what is an essentially a continuous band of (low energy)bonding molecular orbitals and a continuous band of (higher energy) anti-bondingmolecular orbitals. Imagine that instead of two or threeor four atomic orbitals combining to form molecular orbitals, a mole (6 x10 23)atomic orbitals are combined to produce a mole of molecular orbitals. Instead they are dispersedover the whole macroscopic piece of metal. In contrast to H-H or diamond, where the electrons involved in bonding are located(most probably) between the two nuclei, in aluminum and other metals, the valenceelectrons are not closely associated with each nucleus. What emerges is a picture of aluminum nuclei and their core electrons, packed like spheres where one layer of spheres rests in the interstices of the underlying and overlying layers (but remember the uncertainty principle!) With modern microscopes, it is actually quite easy to “see” atoms nowadays, since we can use a variety of techniques to image where the Al atoms are in the solid structure. 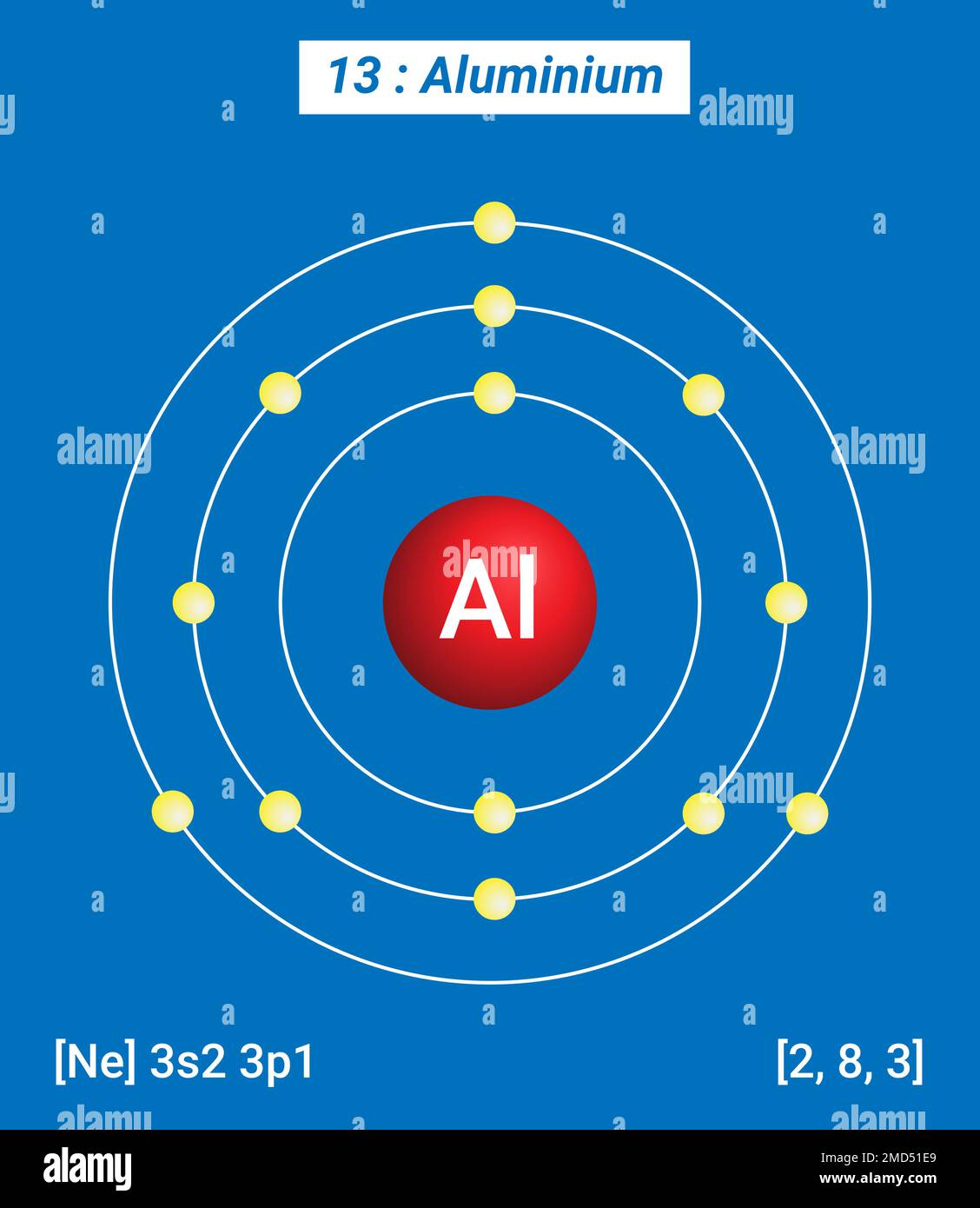
How are atoms arranged in a metal? Let us use aluminum as an example - most of us have something made of aluminum, such as a pan or aluminum foil. All metals conduct electricity, but not all equally. Most are shiny, but not all are colorless - gold and copper have distinct colors. Metals have quite a wide range of properties at normal temperatures, from liquid (like mercury) to extremely hard (like tungsten). Chemistry, life, the universe and everything Chapter 3.4: Metals
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
